 Both bond length and bond energy changes as the bond order increases and as the number of electrons shared between two atoms in a molecule increases, the bond order of a bond increases, the strength of the bond increases and the distance between nuclei decreases (Table 9.10. In general chemistry courses, students learn that covalent bonds can come as either single, double or triple bonds, which are identifies by their bond order. A, and 'X' (see the discussion for O2 in Section 9.1). To explain the observed paramagnetic properties of molecular oxygen with Molecular Orbital theory Example 10.1, Walsh Molecular Orbital Diagrams Using the Walsh MO diagram, Figure 10.5: (a).To describe the connection between bond order, bond length and bond energy in diatomic molecules.This document highlights the progressive lowering of molecular orbital energies due to the the atomic orbital changes, and presents a correlation diagram linking internuclear separation with molecular orbital energies as well.\) There are good diagrams showing the gradual change in energy differences across the second row range of homonuclear diatomic molecules. For period 2 elements, where all the valence electrons of an atom are in s and p orbitals, we find that the Lewis dot structure of molecules will often. More information about the details of this difference can be found in most inorganic chemistry textbooks. On combining these two atomic orbitals of two H-atoms, we get two molecular orbitals for H 2 molecule. Draw the molecular orbital diagrams for the following diatomic molecules/polyatomic ions. 2 (Image) Draw a molecular orbital diagram for triangular H3+ and describe the bonding. In the case of H 2 molecule, each H-atom consists of one atomic orbital (i.e. Construct the molecular orbital diagram for H2 and then identify the bond order. In the dioxygen molecular orbital scheme the s-p mixing effect is no longer significant enough to alter the relative orbital arrangement. According to LCAO method, molecular orbitals are formed by the combination of atomic orbitals. The change of the molecular orbital ordering between nitrogen and oxygen is the manifestation of this decreased s-p mixing. As nuclear charge increases, s-p mixing becomes less significant. The more stabilized 2s orbital does not s-p mix as effectively, due to the greater energy difference between the 2s and 2p orbitals. Molecular Orbital (MO) Diagram for O2 (2-) chemistNATE 2920 Was this helpful Previous video Next video Related Videos 21:36 Molecular Orbital Theory - Bonding & Antibonding MO - Bond Order The Organic Chemistry Tutor 219 04:40 Molecular Orbital (MO) Diagram for O2 (2-) chemistNATE 2920 18:42 The Molecular Orbital Theory. 
This can be seen qualitatively in the first figure here. 
The effective nuclear charge increases to the right of the period, stabilizing the 2s orbital more drastically than the 2p orbital. 
In the dinitrogen molecular orbital scheme, the dashed lines are there to represent s-p mixing influencing the energy of the four molecular orbitals involved. Create a molecular orbital diagram for the B2+ ion and O22+ ion. Be sure your diagram contains all of the electrons in the ion, including any core electrons. This allows sufficient s-p mixing to lower the energy of the σ(2s) and σ*(2s) molecular orbitals, and is energetically offset by an increase in energy of the σ(2p) and σ*(2p) molecular orbitals. Draw the molecular orbital (MO) electron diagram for the B e 2 + 2 molecular ion. The lighter second period elements (prior to oxygen) have a relatively small difference in energy between the 2s and 2p orbitals. S-p mixing is the primary cause of the difference in the molecular orbitals of nitrogen and oxygen, which is influenced by the initial atomic orbital energies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
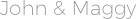
 RSS Feed
RSS Feed
